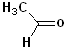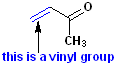Aldehydes & Ketones
(These are a subgroup of the carbonyl family. Aldehydes are characterized by a
Hydrogen atom and a Carbon group attached to the carbonyl. Ketones have the
carbonyl attached to two carbon groups.)
Basic IUPAC nomenclature-ALDEHYDES:
(See the home page for parent root names, prefixes & suffixes.)
1. Number the longest chain of carbon atoms that contains the carbonyl such that
the carbonyl carbon is number 1.
2.
Identify the root name of the
structure; change the suffix from -e to
-al.
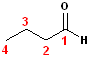 The
root name of the compound is butane, dropping the
-e and changing to -al will give the formal name of
butanal. It is not necessary to use the #1 in the
name; it is understood that the #1 carbon is, by definition the aldehyde carbon.
However, there are two isomers of butanal. This one is the linear or "normal"
isomer.
The
root name of the compound is butane, dropping the
-e and changing to -al will give the formal name of
butanal. It is not necessary to use the #1 in the
name; it is understood that the #1 carbon is, by definition the aldehyde carbon.
However, there are two isomers of butanal. This one is the linear or "normal"
isomer.
There is also a constitutional isomer of butane, which
will have two names-an IUPAC and a common name:
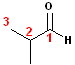 The
aldehyde carbon is still numbered #1, but the carbons are branched to form the
isobutyl group. This isomer has the common name:
isobutyraldehyde. All compounds have an IUPAC name (not all have a common
name) and applying the numbering, as shown, the compound is actually named as a
propan- + -al
which has a methyl group at position #2. 2-Methylpropanal-is
the IUPAC name. Notice, there is no need to use the #1 in the name, since it is
understood that the aldehyde carbon is #1 and the numbering of the chain begins
there.
The
aldehyde carbon is still numbered #1, but the carbons are branched to form the
isobutyl group. This isomer has the common name:
isobutyraldehyde. All compounds have an IUPAC name (not all have a common
name) and applying the numbering, as shown, the compound is actually named as a
propan- + -al
which has a methyl group at position #2. 2-Methylpropanal-is
the IUPAC name. Notice, there is no need to use the #1 in the name, since it is
understood that the aldehyde carbon is #1 and the numbering of the chain begins
there.
3. If the
compound contains a double bond i.e. it is also an alkene, then change the infix
from -an- to -en-
and the placement of the infix is determined by the numbering.
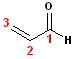 The 2
Carbon base name of this compound is propane. There are two functional groups
present and the name must account for both of them, and a C=O takes precedent
over the C=C. So, the base name becomes: propanal.
The aldehyde carbon defines position #1; the alkene begins at C#2; the name is:
2-Propenal.
The 2
Carbon base name of this compound is propane. There are two functional groups
present and the name must account for both of them, and a C=O takes precedent
over the C=C. So, the base name becomes: propanal.
The aldehyde carbon defines position #1; the alkene begins at C#2; the name is:
2-Propenal.
This is a combination frequently found in organic
chemistry: the carbonyl and the alkene. It is usually referred to as an
a,b-unsaturated
carbonyl compound.
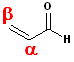 The carbon
atom connected to the carbonyl is designated the alpha (a)
carbon and the second carbon of the double bond is the beta (b)
carbon. Thus, the double bond, linking the two carbons is called: a,b.
However, the #1 carbon in the system, is still the carbonyl
carbon.
The carbon
atom connected to the carbonyl is designated the alpha (a)
carbon and the second carbon of the double bond is the beta (b)
carbon. Thus, the double bond, linking the two carbons is called: a,b.
However, the #1 carbon in the system, is still the carbonyl
carbon.
4.
When the aldehyde functional
group is attached to a ring, the entire suffix:-carbaldehyde
is used. Examples are:
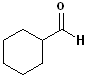 |
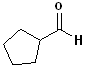 |
|
Cyclohexanecarbaldehyde |
Cyclopentanecarbaldehyde |
5.
The IUPAC system retains several common names:
|
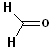
|
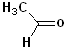
|
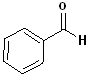 |
|
| Formaldehyde (derived from formic
acid: HCOOH) |
Acetaldehyde (derived from acetic
acid H3CCOOH) |
Benzaldehyde |
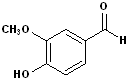
Vanillin |
Basic IUPAC nomenclature-KETONES:
(See the home page for parent root names, prefixes & suffixes.)
1. Number the longest chain of carbon atoms that contains the carbonyl carbon,
such that the C=O has the smallest number.
2.
Change the -e of the alkane to
-one.
3.
Cyclic compounds are named in
the same fashion; e.g. cyclohexan- +
-one = cyclohexanone.
4. The IUPAC system retains several common names:
 |
 |
 |
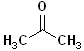
| Acetone (propanone) |
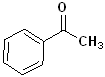
Acetophenone
|
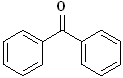
Benzophenone |
| |
|
|
Common Names
for Aldehydes & Ketones
1. For aldehydes,
common names are derived from their corresponding carboxylic acids:
Formaldehyde (derived from formic acid HCOOH);
Acetaldehyde (derived from acetic acid H3CCOOH)
2.
For ketones, the two alkyl
groups are named, alphabetized and placed in front of the term "ketone":
 |
 |
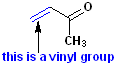 |
 |
 |
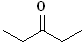
| Diethyl ketone |
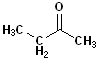
Ethyl methyl ketone |
Methyl vinyl ketone
(MVK) |
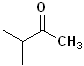
Dimethyl ketone |
Methyl isopropyl ketone |
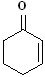
EXERCISE: Write the IUPAC name for each
of the following structures.
| Structure |
Name |
Structure |
Name |
| 1 |
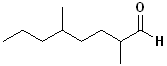 |
 |
6 |
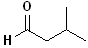 |
 |
| 2 |
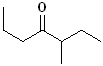 |
 |
7 |
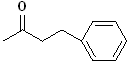 |
 |
| 3 |
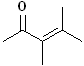 |
 |
8 |
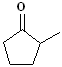 |
 |
| 4 |
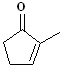 |
 |
9 |
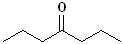 |
 |
| 5 |
 |
 |
10 |
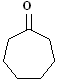 |
 |
END OF SECTION
email questions & comments to:
Dr. JA Colapret
 The
root name of the compound is butane, dropping the
-e and changing to -al will give the formal name of
butanal. It is not necessary to use the #1 in the
name; it is understood that the #1 carbon is, by definition the aldehyde carbon.
However, there are two isomers of butanal. This one is the linear or "normal"
isomer.
The
root name of the compound is butane, dropping the
-e and changing to -al will give the formal name of
butanal. It is not necessary to use the #1 in the
name; it is understood that the #1 carbon is, by definition the aldehyde carbon.
However, there are two isomers of butanal. This one is the linear or "normal"
isomer. The
aldehyde carbon is still numbered #1, but the carbons are branched to form the
isobutyl group. This isomer has the common name:
isobutyraldehyde. All compounds have an IUPAC name (not all have a common
name) and applying the numbering, as shown, the compound is actually named as a
propan- + -al
which has a methyl group at position #2. 2-Methylpropanal-is
the IUPAC name. Notice, there is no need to use the #1 in the name, since it is
understood that the aldehyde carbon is #1 and the numbering of the chain begins
there.
The
aldehyde carbon is still numbered #1, but the carbons are branched to form the
isobutyl group. This isomer has the common name:
isobutyraldehyde. All compounds have an IUPAC name (not all have a common
name) and applying the numbering, as shown, the compound is actually named as a
propan- + -al
which has a methyl group at position #2. 2-Methylpropanal-is
the IUPAC name. Notice, there is no need to use the #1 in the name, since it is
understood that the aldehyde carbon is #1 and the numbering of the chain begins
there. The 2
Carbon base name of this compound is propane. There are two functional groups
present and the name must account for both of them, and a C=O takes precedent
over the C=C. So, the base name becomes: propanal.
The aldehyde carbon defines position #1; the alkene begins at C#2; the name is:
2-Propenal.
The 2
Carbon base name of this compound is propane. There are two functional groups
present and the name must account for both of them, and a C=O takes precedent
over the C=C. So, the base name becomes: propanal.
The aldehyde carbon defines position #1; the alkene begins at C#2; the name is:
2-Propenal. The carbon
atom connected to the carbonyl is designated the alpha (a)
carbon and the second carbon of the double bond is the beta (b)
carbon. Thus, the double bond, linking the two carbons is called: a,b.
However, the #1 carbon in the system, is still the carbonyl
carbon.
The carbon
atom connected to the carbonyl is designated the alpha (a)
carbon and the second carbon of the double bond is the beta (b)
carbon. Thus, the double bond, linking the two carbons is called: a,b.
However, the #1 carbon in the system, is still the carbonyl
carbon.