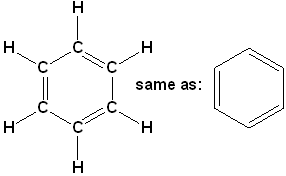
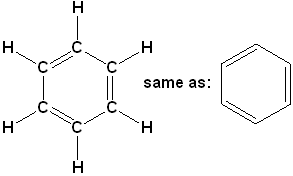
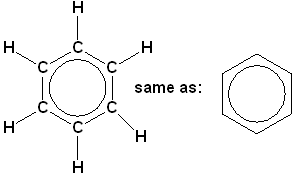
Expanded structural formula with condensed line angle formula.
This representation is not as common in chemistry references, but is often found in biological & biochemistry.
Benzene & Its Derivatives
(The class of compounds, which contains benzene or substituted derivative of benzene)
Benzene
There is a class of hydrocarbons, that is characterized by a high degree of unsaturation and unusual stability. The most important and common member of this class is benzene (C6H6). The formula is represented in several ways:
|
|
|
|
|
Expanded structural formula with condensed line angle formula. |
This representation is not as common in chemistry references, but is often found in biological & biochemistry. |
|
Monosubstituted Benzene
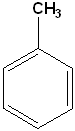
When one of the positions on the ring has been substituted with another atom or group of atoms, the compound is a monosubstituted benzene. If two positions are replaced, then it's a disubstituted, & so on. Many of the monosubstituted compounds have their own name and some of the more common ones are listed in the Table below:
| HYDROCARBONS | |||
 |
 |
 |
 |
| Toluene (Methylbenzene) |
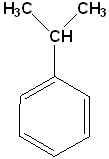
Cumene (Isopropylbenzene) |
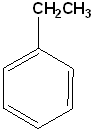
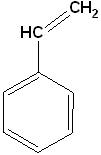
Ethylbenzene | Styrene (Vinylbenzene) |
| OTHER FUNCTIONAL GROUPS | |||
 |
 |
 |
 |
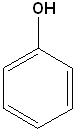
| Phenol (Hydroxybenzene) |
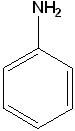
Aniline (Aminobenzene) |
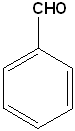
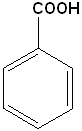
Benzaldehyde | Benzoic Acid |
 |
 |
 |
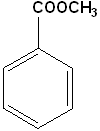 |
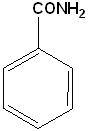
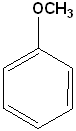
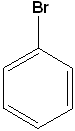
| Benzamide | Anisole | Bromobenzene | Methylbenzoate |
Disubstituted Benzenes
When two of the positions on the ring has been substituted with another atom or group of atoms, the compound is a disubstituted benzene. There is a separate nomenclature to describe the relative positions. Using toluene as the example, the ortho orientation is the 1,2 relationship; the meta is 1,3 and the para is 1,4. Note that there are two ortho and meta positions, but only one para.
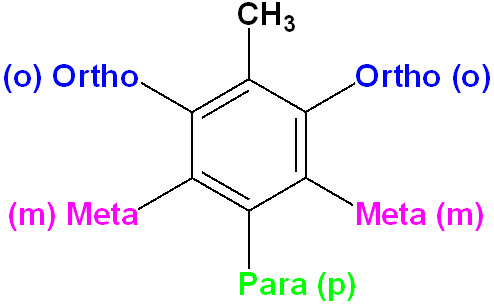
Where one group imparts a special name, name the compound as a derivative of that molecule:
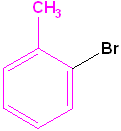 |
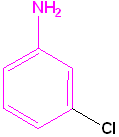 |
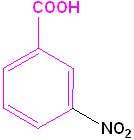 |
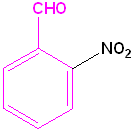 |
| Toluene is the parent name | Aniline is the parent name | Benzoic acid is the parent name | Benzaldehyde is the parent name |
| 2-Bromotoluene | 3-Chloroaniline | 3-Nitrobenzoic acid | 2-Nitrobenzaldehyde |
| o-Bromotoluene | m-Chloroaniline | m-Nitrobenzoic acid | o-Nitrobenzaldehyde |
If neither group imparts a special name, locate the groups & alphabetize.
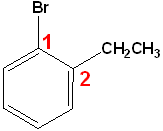 |
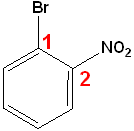 |
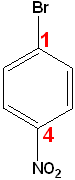 |
| 1-Bromo-2-ethylbenzene | 1-Bromo-2-nitrobenzene | 1-Bromo-4-nitrobenzene |
| o-Bromoethylbenzene | o-Bromonitrobenzene | p-Bromonitrobenzene |
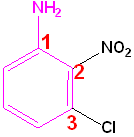
Polysubstituted Benzenes
If one group imparts a special name, name the molecule as a derivative of that compound and if no group imparts a special name, list them in alphabetical order, giving them the lowest set of numbers
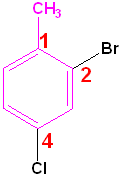 |
 |
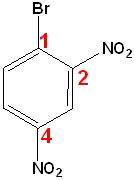 |
| Toluene is the parent name | Aniline is the parent name | No special name |
| 2-Bromo-4-chlorotoluene | 3-Chloro-2-nitroaniline | 1-Bromo-2,4-dinitrobenzene |
EXERCISE: Write the IUPAC name for each of the following structures.
| Structure | Name | Structure | Name | ||
| 1 | 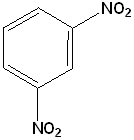 |
|
11 | 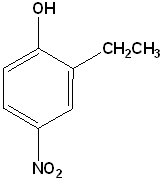 |
|
| 2 | 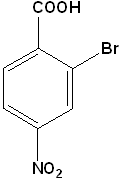 |
|
12 | 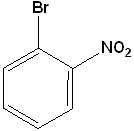 |
|
| 3 | 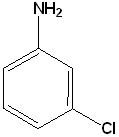 |
|
13 | 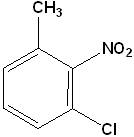 |
|
| 4 | 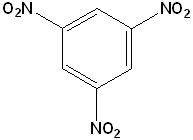 |
|
14 | 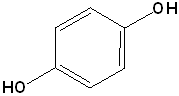 |
|
| 5 | 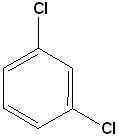 |
|
15 | 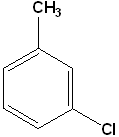 |
|
| 6 | 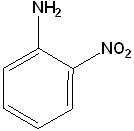 |
|
16 | 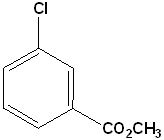 |
|
| 7 | 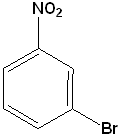 |
|
17 | 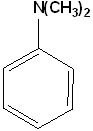 |
|
| 8 | 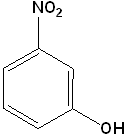 |
|
18 | 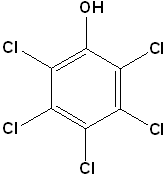 |
|
| 9 | 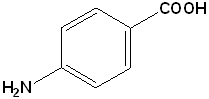 |
|
19 | 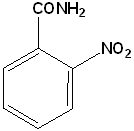 |
|
| 10 | 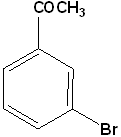 |
|
20 | 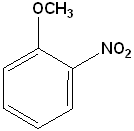 |
|
END OF SECTION
email questions & comments to: Dr. JA Colapret